Quality
We never compromise on excellence.


We never compromise on excellence.
We embrace technology to push boundaries.
We move quickly and deliver on time, every time.
We care about the people behind every project.

Valuable experience from 54 successful NDA/BLAs directly converted into AI training data, ensuring model professionalism and practicality.
Deep integration of biostatistics and AI technology, understanding both business and technology, avoiding "AI for AI's sake" pitfalls.
All solutions comply with 21 CFR Part 11, GDPR, and other regulatory requirements, eliminating compliance risks for clients.
Existing AI Dashboard system and Agent development capabilities enable rapid deployment of new services, reducing implementation risks.
Supporting cloud, on-premise, and hybrid deployment models to meet diverse client security and cost requirements.

An industry-leading data labeling system powered by 15+ years of clinical trial expertise and AI technology.

A comprehensive knowledge network built on 50+ successfully approved NDA/BLA projects, covering disease–drug–gene–endpoint relationships.

AI continuously refined by 200+ biostatisticians and data management experts, ensuring professional-grade outputs through real-world human feedback.

A clinical development-specific intelligent Q&A system built on ICH guidelines, FDA/EMA requirements, and 1,800+ historical trial datasets.

End-to-end process automation covering the complete chain from EDC data cleaning and quality checks to TFL generation.

Modular, plug-and-play AI function libraries for rapid development of custom clinical decision support tools — powered by 6 patented technologies and a 50-member AI R&D team.

Advanced biometrics integrating clinical intelligence with automation to deliver submission-ready data with precision and uncompromising integrity.

Streamline your clinical data lifecycle with our AI-enhanced EDC&CDM solutions. From database design and eCRF development to medical coding and accelerated database lock, we reduce queries by 60% while ensuring 100% data integrity. Platform-agnostic expertise across all major EDC systems delivers seamless, compliant study execution.
View Details
Full-service biostatistics and CDISC programming from study design to submission. Expert teams deliver SAP, SDTM/ADaM and TFLs with double-programming QC. AI-enhanced workflows reduce timelines by 40% while maintaining 100% regulatory compliance.
View Details
Accelerate drug development with advanced PK/PD modeling and AI-enhanced analytics. Comprehensive services from non compartmental analysis through population modeling to dosing optimization. IND to NDA support delivering regulatory-grade documentation for FDA/EMA/PMDA and more regulatory agencies.
View DetailsTransforming complex data into actionable insights through AI-powered analytics to optimize trial oversight and decision-making.
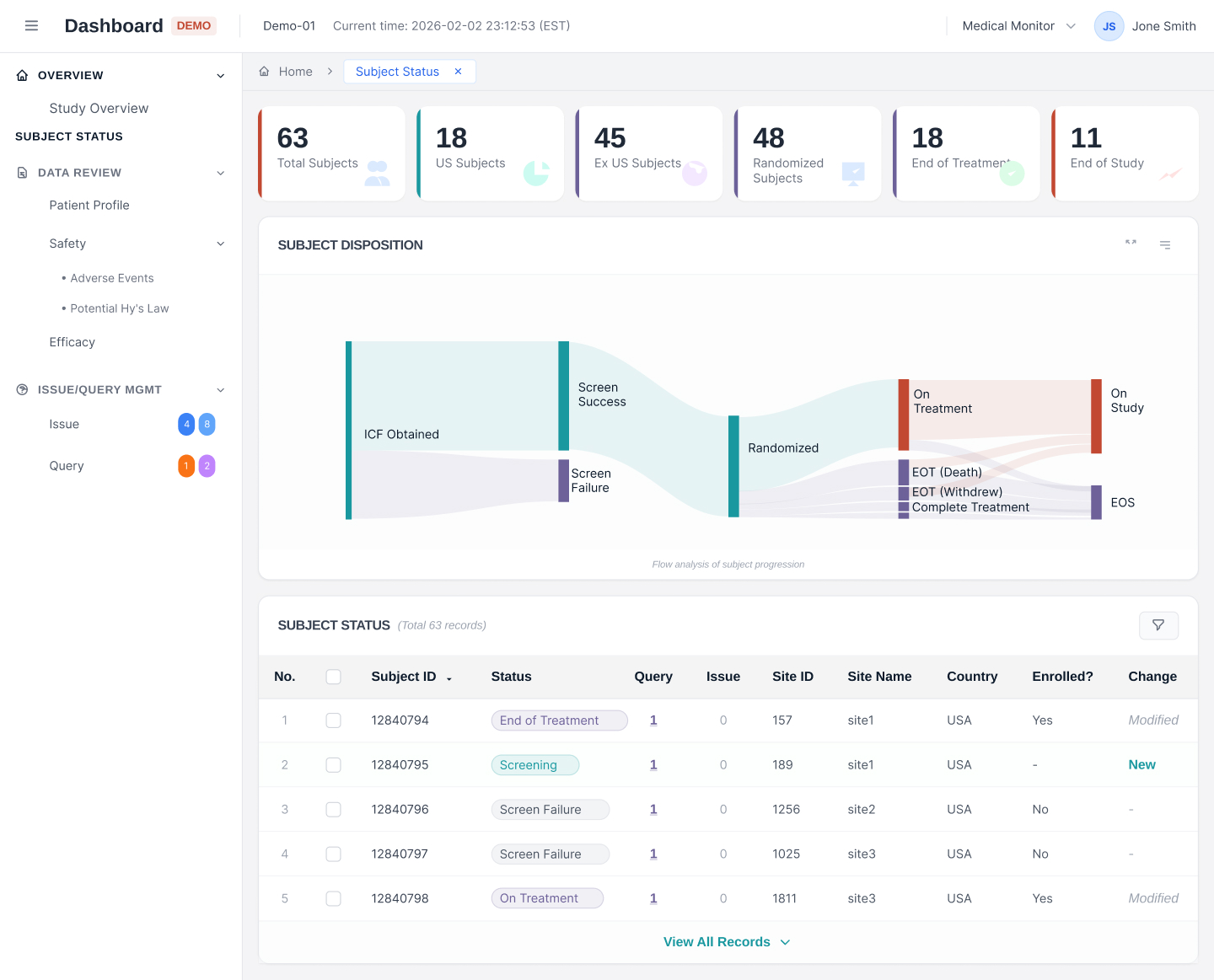
Real-time clinical trial monitoring with interactive dashboards. Track enrollment, safety signals, and data quality metrics at a glance. Customizable views enable sponsors and CRAs to make data-driven decisions faster with intuitive visualizations and automated alerts.
View Details
AI-powered automation for routine data management tasks. Our intelligent agent handles medical coding suggestions, query generation, and data reconciliation, reducing manual effort by up to 60% while maintaining regulatory compliance and audit trails.
View Details
Tailored AI solutions for your unique clinical development challenges. From predictive analytics for patient recruitment to automated safety signal detection, we build custom models that integrate seamlessly with your existing workflows and systems.
View Details

Comprehensive medical affairs support from protocol development through post-marketing. Our team of MDs and medical writers ensures scientific accuracy, regulatory alignment, and clear communication across all clinical documentation and stakeholder interactions.


We never compromise on excellence.
We embrace technology to push boundaries.
We move quickly and deliver on time, every time.
We care about the people behind every project.


Valuable experience from 54 successful NDA/BLAs directly converted into AI training data, ensuring model professionalism and practicality.
Deep integration of biostatistics and AI technology, understanding both business and technology, avoiding "AI for AI's sake" pitfalls.
All solutions comply with 21 CFR Part 11, GDPR, and other regulatory requirements, eliminating compliance risks for clients.
Existing AI Dashboard system and Agent development capabilities enable rapid deployment of new services, reducing implementation risks.
Supporting cloud, on-premise, and hybrid deployment models to meet diverse client security and cost requirements.
An industry-leading data labeling system powered by 15+ years of clinical trial expertise and AI technology.


A comprehensive knowledge network built on 50+ successfully approved NDA/BLA projects, covering disease–drug–gene–endpoint relationships.


AI continuously refined by 200+ biostatisticians and data management experts, ensuring professional-grade outputs through real-world human feedback.


A clinical development-specific intelligent Q&A system built on ICH guidelines, FDA/EMA requirements, and 1,800+ historical trial datasets.


End-to-end process automation covering the complete chain from EDC data cleaning and quality checks to TFL generation.


Modular, plug-and-play AI function libraries for rapid development of custom clinical decision support tools — powered by 6 patented technologies and a 50-member AI R&D team.



Advanced biometrics integrating clinical intelligence with automation to deliver submission-ready data with precision and uncompromising integrity.


AI-Enhanced CDM
Streamline your clinical data lifecycle with our AI-enhanced EDC&CDM solutions. From database design and eCRF development to medical coding and accelerated database lock, we reduce queries by 60% while ensuring 100% data integrity. Platform-agnostic expertise across all major EDC systems delivers seamless, compliant study execution.
Learn More

Accelerated Data Insights
Full-service biostatistics and CDISC programming from study design to submission. Expert teams deliver SAP, SDTM/ADaM and TFLs with double-programming QC. AI-enhanced workflows reduce timelines by 40% while maintaining 100% regulatory compliance.
Learn More

Advanced PK/PD Analytics
Accelerate drug development with advanced PK/PD modeling and AI-enhanced analytics. Comprehensive services from non compartmental analysis through population modeling to dosing optimization. IND to NDA support delivering regulatory-grade documentation for FDA/EMA/PMDA and more regulatory agencies.
Learn More

Real-Time Trial Insights
Real-time clinical trial monitoring with interactive dashboards. Track enrollment, safety signals, and data quality metrics at a glance. Customizable views enable sponsors and CRAs to make data-driven decisions faster with intuitive visualizations and automated alerts.
Learn More

Intelligent Data Automation
AI-powered automation for routine data management tasks. Our intelligent agent handles medical coding suggestions, query generation, and data reconciliation, reducing manual effort by up to 60% while maintaining regulatory compliance and audit trails.
Learn More

Tailored AI Models
Tailored AI solutions for your unique clinical development challenges. From predictive analytics for patient recruitment to automated safety signal detection, we build custom models that integrate seamlessly with your existing workflows and systems.
Learn More

Comprehensive medical affairs support from protocol development through post-marketing. Our team of MDs and medical writers ensures scientific accuracy, regulatory alignment, and clear communication across all clinical documentation and stakeholder interactions.

Accelerating Drug Development with Data-Driven Biometrics & Clinical Intelligence.
Submit RFP